Chiesi’s values have always been integrated in our business model. Our work and our commitment are driven by the goal of improving people quality of human life, putting people at the centre of our actions, being them our patients or our collaborators.

Chiesi’s people are more than 5.500 all over the world.
Everyday, we try guarantee our employees a familiar, safe and stimulating working environment. We focus on our people’s well-being, with programs dedicated to People Development and People Care.
Among these programs:

Chiesi works in four therapeutic areas:
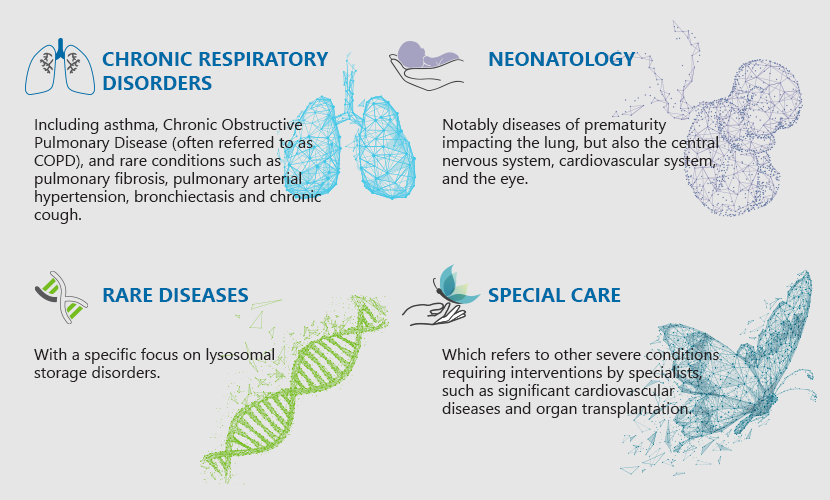
The responsibility toward our patients translate into quality and integrity of our products, listening, comprehension of needs and access to care.
More than 21% of our turnover is dedicated to Research and Development and our work is based putting the patient at the centre, investigating on innovative therapeutic solutions which are able to improve the quality of care and the quality of people and caregivers’ life.
To promote health as a fundamental human right, we are committed to expand the access to care for those patients who are in difficult situations and are unable to achieve access to care.
For that purpose, we work together with associations and we promote programs of early access for our medicines, with a focus on exclusive Orphan Medicinal Products with high unmet needs and no alternative treatments available (read here our policy)
To discover more about responsibility toward our people and our patients, click here to read our Sustainability Report.